Description
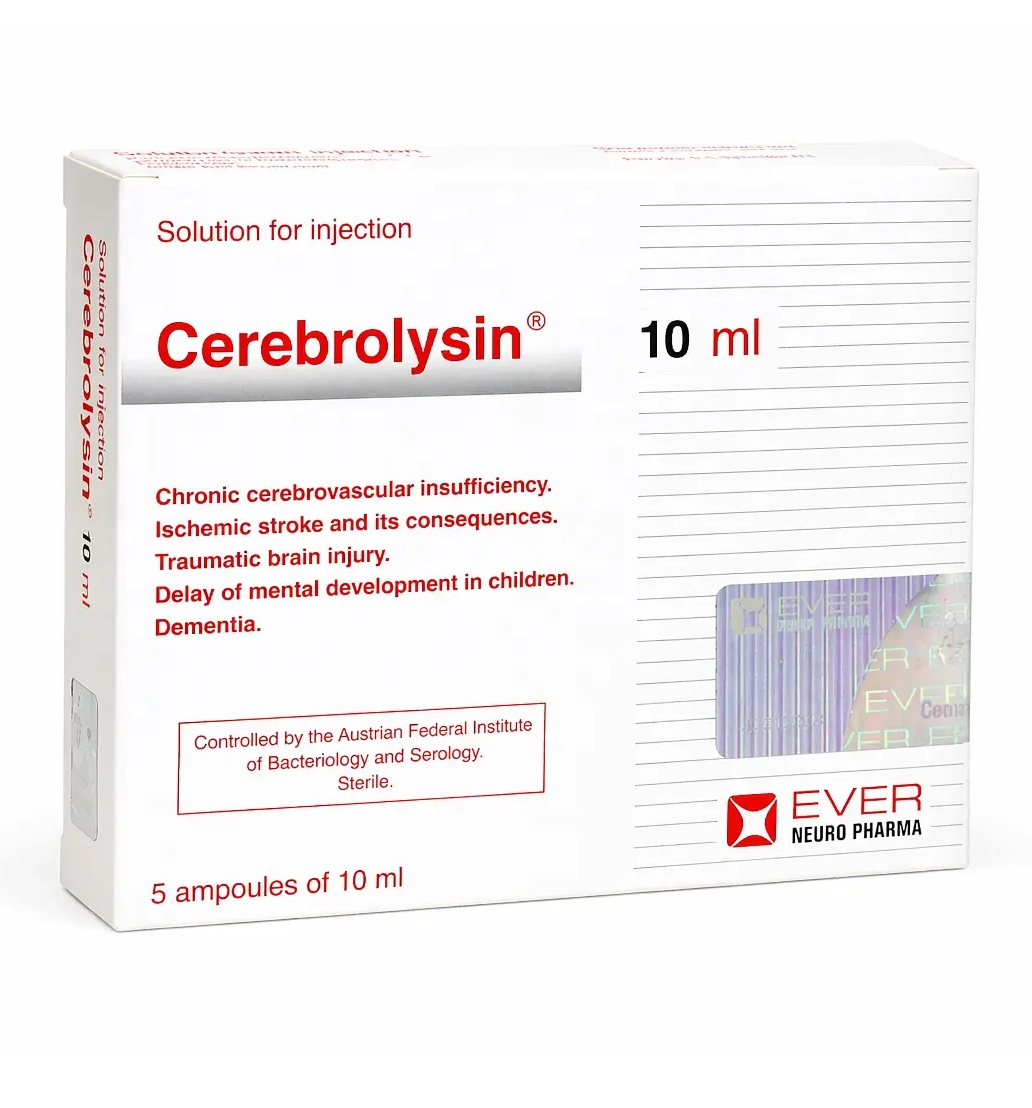
Cerebrolysin Solution 215.2 mg/ml, 10 ml Ampoules, No. 5
Cerebrolysin Solution 215.2 mg/ml is a peptide-based injectable medicine formulated as a sterile solution for parenteral use. It is widely known as a multimodal neuropeptide preparation that supports neurological recovery, brain metabolism, and the functional restoration of nervous tissue in clinical settings where neurotrophic support is relevant.
This presentation comes in 10 ml ampoules, No. 5, meaning the package contains five ampoules, each with a 10 ml volume. At a concentration of 215.2 mg/ml, each ampoule contains a substantial total amount of active peptide complex, which is one reason Cerebrolysin is typically used under medical supervision and as part of structured treatment protocols rather than as a casual wellness product.
Cerebrolysin is commonly discussed in the context of stroke rehabilitation, traumatic brain injury, vascular cognitive decline, dementia support, and other neurological conditions where restoration of neuronal function and protection from secondary injury are important. It is not a simple symptomatic medicine. Instead, it is generally described as a biologically active peptide mixture with neurotrophic and neuroprotective properties.
Is Cerebrolysin Useful in Psychiatry Disorders? (PMC)
Composition and Dosage Form
- Active substance: Cerebrolysin, a peptide mixture
- Concentration: 215.2 mg/ml
- Volume of each ampoule: 10 ml
- Total amount per ampoule: 2,152 mg
- Package: 5 ampoules
- Dosage form: Sterile solution for injection / infusion
The injectable solution form is especially important because peptide-based medicines are not usually suitable for oral use in the same way as standard tablets or capsules. By being supplied as a sterile ampoule solution, Cerebrolysin is prepared for professional parenteral administration, which allows direct delivery into the bloodstream or muscle according to approved protocols in the relevant country.
The ampoule format also supports accurate dosing. In neurological practice, precision matters because treatment goals are often tied to recovery phases, rehabilitation programs, or adjunctive therapy plans. A five-ampoule package is practical for short courses, initiation phases, or controlled treatment schedules.
Pharmacological Classification
- Pharmacological group: Neurotrophic / nootropic / neuroprotective agent
- Class: Multimodal peptide preparation
- Mechanism type: Neuroregenerative support, trophic modulation, and cerebral metabolism support
Cerebrolysin is usually described as a complex neuropeptide drug derived from enzymatically processed brain proteins. In practical pharmacological language, it is not a classic small-molecule sedative, analgesic, or stimulant. Instead, it belongs to the broader category of agents intended to support neuronal resilience, synaptic function, and recovery after central nervous system injury or dysfunction.
The word “multimodal” is important because the medicine is discussed as acting through more than one biological pathway. This includes potential effects on neurotrophic signaling, neuronal survival, synaptic plasticity, and anti-excitotoxic support. As a result, Cerebrolysin has become a recognizable brand in neurological and rehabilitation-oriented medicine markets.
What Cerebrolysin Is Commonly Used For
Cerebrolysin is most often associated with conditions in which the brain has been affected by injury, ischemia, degeneration, or prolonged functional decline. Its use depends on country-specific approval and clinical judgment, but the medicine is commonly discussed in relation to:
- Stroke rehabilitation and post-stroke recovery
- Traumatic brain injury
- Cognitive impairment and vascular cognitive decline
- Alzheimer-type and mixed dementia support
- Neurological recovery after brain insult
- Adjunctive support in selected neuropsychiatric contexts
For an informational resource, it is important to be accurate: Cerebrolysin is not a universal cure for neurological disease. Instead, it is a specialized therapy used to support recovery mechanisms in contexts where brain function is compromised. Its reputation comes from its neurotrophic profile, not from broad symptomatic suppression.
How the Medicine Works
Cerebrolysin is described as a peptide preparation with neurotrophic-like properties. In plain language, this means it may help support the biological environment that neurons need in order to survive, recover, and communicate more effectively. The medicine is often discussed in relation to endogenous repair mechanisms, which are the body’s own internal processes for restoration after injury or stress.
Its proposed mechanisms include:
- Support of neuronal survival and repair pathways
- Modulation of synaptic plasticity
- Improvement of cerebral metabolic function
- Reduction of secondary damage after neuronal injury
- Support of neurotrophic signaling pathways
The exact pharmacology is complex. Cerebrolysin is not a single molecule with one receptor target. It is a mixture of low-molecular-weight peptides and amino acids that may influence several pathways at once. That is why it is called multimodal. From a clinical point of view, this can be useful in conditions where the brain needs broad support rather than narrow receptor blockade.
Another important concept is that this medicine is used as a supportive agent. It may be included in treatment plans where rehabilitation, prevention of further decline, and functional recovery are major goals. This makes it especially relevant in neurology, geriatrics, and neurorehabilitation.
Why Cerebrolysin Attracts Scientific Interest
Cerebrolysin attracts ongoing attention because it sits at the intersection of neurobiology, rehabilitation medicine, and peptide pharmacology. Many standard neurological drugs reduce symptoms, but they do not necessarily focus on tissue recovery. Cerebrolysin is interesting because it is discussed as a medicine that may support repair-oriented processes in the brain.
This has led to research interest in areas such as:
- Acute and subacute stroke recovery
- Chronic cognitive impairment
- Neuroprotection after traumatic injury
- Adjunctive treatment in selected psychiatric contexts
- Enhancement of rehabilitation outcomes
Scientific discussion also reflects a practical reality: results are mixed across different studies, patient groups, and treatment phases. That does not make the medicine irrelevant. It means the evidence should be described carefully and responsibly. A strong product description should present Cerebrolysin as a legitimate medical preparation with clinical use in some regions, while also acknowledging that the magnitude of benefit can vary.
For readers who want to explore the research background, the following scientific source is useful:
Cerebrolysin for acute ischaemic stroke (Cochrane review)
Clinical Contexts and Therapeutic Relevance
Cerebrolysin is often used in settings where recovery is time-sensitive and neurological function needs structured support. That is one reason it remains a known product in neurology and rehabilitation clinics in several countries. In stroke care, for example, the medicine is frequently discussed as part of an adjunctive strategy rather than as a stand-alone intervention.
Its practical relevance may be described in several ways:
- Support during post-acute neurological recovery
- Adjunctive use alongside rehabilitation and physiotherapy
- Potential support for cognitive performance in selected patients
- Application in patients with reduced brain resilience after injury or ischemia
In dementia-related discussions, Cerebrolysin is often considered because of its neurotrophic profile and possible influence on cognition and daily functioning. In traumatic brain injury, the rationale is similar: the brain may benefit from agents that support recovery pathways rather than simply suppress symptoms.
Administration and Use
Cerebrolysin is a parenteral medicine and should be administered according to local prescribing information and professional guidance.
- Route: Injection or infusion, depending on the protocol
- Preparation: Use only as directed in the official leaflet or by a healthcare professional
- Setting: Medical supervision is recommended
- Course structure: Often used in scheduled treatment courses
Because it is an injectable peptide solution, correct handling is important. The medicine should not be treated like an over-the-counter oral supplement. Dosage, speed of administration, dilution requirements, and treatment duration may vary according to diagnosis, age, clinical context, and regional labeling. That variability is one reason why this medicine is commonly associated with specialist care.
For readers looking for practical information, the key message is simple: Cerebrolysin is a professional-use injectable neurological medicine, not a self-selected consumer product.
Possible Benefits in Clinical Practice
Cerebrolysin is often described in clinical discussions as a medicine that may contribute to better recovery dynamics. While the strength of evidence varies by indication, the intended benefits are usually tied to overall neurological support rather than instant symptom control.
Commonly discussed benefits include:
- Support of post-injury brain recovery
- Potential improvement in cognitive performance
- Assistance in rehabilitation-oriented treatment plans
- Neurotrophic and neuroprotective support
- Possible benefit in specific patient groups with neurological compromise
These points are best framed as “potential” or “reported” benefits because the medicine’s clinical value can depend on disease stage, treatment setting, and the quality of the supporting studies. In SEO copy, that careful wording increases trust and reduces the risk of overclaiming.
Scientific Background and Evidence Perspective
The scientific literature on Cerebrolysin includes studies in stroke, dementia, traumatic brain injury, and selected psychiatric disorders. Some publications suggest supportive effects on cognition or recovery, while others note that more high-quality evidence is needed. This mixed evidence profile is common for complex neurobiological agents.
One recent review focused on psychiatric disorders notes that Cerebrolysin has been used for years mainly in neurological practice and may have a limited but possibly meaningful role in certain mental health contexts. That kind of literature is useful because it shows how the medicine is being reconsidered beyond traditional neurology alone.
If you want a second scientific reference to support a broader understanding of the medicine, this review is relevant:
Is Cerebrolysin Useful in Psychiatry Disorders? (PMC)
The value of these sources is not to imply a universal indication, but to show that Cerebrolysin remains a studied neuroactive preparation with continuing research interest. For a content page, this supports a balanced narrative: biologically active, clinically used in some regions, studied in several neurological conditions, and still the subject of evidence discussion.
Safety and Tolerability
As with any injectable medicine, safety depends on the patient, the indication, the route of administration, and adherence to professional instructions. Cerebrolysin is generally discussed as a medicine with a manageable tolerability profile in many clinical contexts, but side effects can occur.
Possible unwanted effects may include:
- Dizziness
- Headache
- Nausea
- Feeling hot or flushed
- Injection-related discomfort
- Other temporary adverse reactions in sensitive patients
Because the medicine is administered parenterally, practical safety also includes sterile technique, correct dilution when needed, and proper observation during treatment. People with a history of hypersensitivity to components of the formulation should avoid use unless advised otherwise by a physician. Patients with epilepsy, significant kidney disease, or complicated medical histories should only use the medicine under direct professional supervision if it is considered appropriate.
For a WordPress medicine page, it is best to present safety in a sober and non-alarmist way. The goal is to inform, not to frighten. At the same time, avoiding unrealistic promises is essential.
Storage Conditions
- Store according to the manufacturer’s instructions
- Protect from excessive heat
- Keep away from direct light
- Do not freeze unless the label specifically allows it
- Keep out of reach of children
Because Cerebrolysin is supplied as a sterile injectable solution, storage conditions are important for maintaining quality and safety. Ampoules should remain intact until use, and any opened or improperly stored product should not be used. If the pack is intended for hospital or clinic use, the responsibility for correct handling is even more important.
Frequently Asked Questions
Is Cerebrolysin a tablet or capsule?
No. Cerebrolysin in this presentation is an injectable solution supplied in ampoules.
What does 215.2 mg/ml mean?
It means that each milliliter of solution contains 215.2 mg of the active peptide mixture.
How much is in one 10 ml ampoule?
Each ampoule contains 2,152 mg of active substance in total.
What does No. 5 mean?
It means the package contains five ampoules.
What is Cerebrolysin mainly used for?
It is mainly discussed in neurological recovery, stroke rehabilitation, cognitive impairment, and traumatic brain injury support, depending on country-specific labeling.
Is Cerebrolysin an everyday supplement?
No. It is a medical injectable preparation and should be used under professional guidance.
SEO-Oriented Summary
Cerebrolysin Solution 215.2 mg/ml, 10 ml Ampoules, No. 5 is a peptide-based injectable medicine widely associated with neurological recovery, neuroprotection, and neurotrophic support. It is described as a multimodal neuropeptide preparation that may be used in clinical settings involving stroke rehabilitation, traumatic brain injury, cognitive decline, and selected neuropsychiatric contexts. Its sterile ampoule form, high concentration, and structured packaging make it a recognizable option in neurology-focused pharmaceutical descriptions.
For SEO purposes, this page naturally covers major search terms such as Cerebrolysin injection, Cerebrolysin 215.2 mg/ml, Cerebrolysin 10 ml ampoules, neurotrophic peptide, brain recovery support, stroke rehabilitation medicine, and injectable nootropic therapy. The most accurate way to describe it is as a professional-use neurological medicine with a research background and a multimodal mechanism profile.
Conclusion
Cerebrolysin 215.2 mg/ml solution, 10 ml ampoules No. 5 is a specialized injectable peptide medicine designed for clinical use in neurological practice. Its value lies in its neurotrophic, neuroprotective, and recovery-supporting profile, which makes it relevant in treatment strategies where brain function, rehabilitation, and cognitive support are important.
The product is best understood as a multimodal parenteral therapy rather than a routine symptomatic drug. Its ampoule format supports accurate dosing and professional administration, while the scientific literature continues to explore where and how this medicine may be most useful. For readers and search engines alike, Cerebrolysin remains one of the better-known peptide-based neurological preparations in modern pharmaceutical use.

Reviews
There are no reviews yet.